Results are being presented today at the International Conference on Brain Tumor Research and Therapy in Norway and published in The New England Journal of Medicine.

A phase 1 trial of a therapy developed by Duke Neurosurgery researchers shows significantly improved long-term survival for patients with recurrent glioblastoma. The poliovirus therapy showed a three-year survival benefit of 21 percent. In comparison, just 4 percent of patients at Duke with the same type of brain tumors were alive at three years when undergoing the standard treatment.
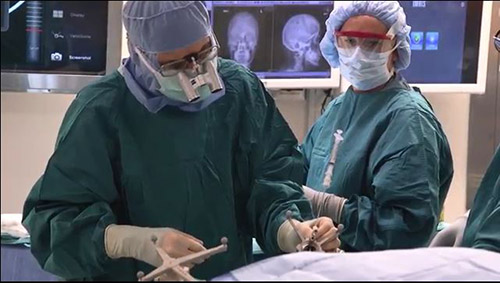
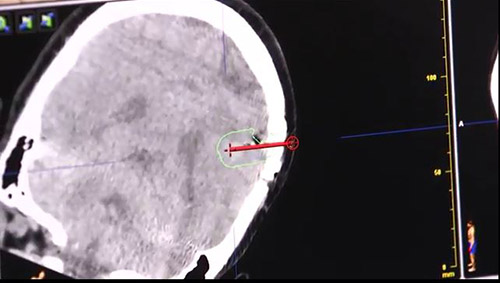
The therapy involves infusing the poliovirus vaccine directly into the brain tumor through a surgically implanted catheter. Developed by Duke’s Matthias Gromeier, MD, the genetically modified virus targets the tumor cells, creating an immune response.
“With the survival rates in this early phase of the poliovirus therapy, we are encouraged and eager to continue with the additional studies that are already under way or planned,” said Darell Bigner, MD, PhD, senior author of the study.
Director of the Preston Robert Tisch Brain Tumor Center at Duke and co-senior author David Ashley, PhD, MBBS, along with co-lead authors Gromeier and Annick Desjardins, MD, reported median follow-up of 27.6 months in the phase 1 trial, which was launched in 2012 with a young patient who is now in remission and working as a pediatric oncology nurse.
In 2016, the poliovirus therapy obtained “breakthrough therapy” designation from the U.S. Food and Drug Administration. Duke Neurosurgery began this year to test the therapy in pediatric brain tumors. Some breast cancer and melanoma patients will soon be eligible to join clinical trials that expand the therapy beyond brain tumors.
For all 61 patients in the study, the median overall survival was 12.5 months, compared to 11.3 months for the historical control group. Starting at two years after treatment, the survival curves in the two groups diverged. The rate of overall survival of poliovirus patients at 24 months was 21 percent, compared to 14 percent for the historical controls. At three years, the gap widened further, with a survival rate of 21 percent for poliovirus patients, compared to 4 percent in the control group. Study participants were selected based on the size and location of their tumor and other factors. The comparison group of patients was drawn from historical cases at Duke involving patients who would have matched the poliovirus enrollment criteria.
Not everyone responds to the poliovirus therapy, but those that do often become long-term survivors, Desjardins says. “The big question is, how can we make sure that everybody responds?”
An 18-year collaboration with the National Cancer Institute and the Food and Drug Administration enabled the pre-clinical and translational phases of development of the new therapy. “This promising approach in glioblastoma therapy exemplifies the strategic investment made by the NCI to support the development of new therapies such as this one from research discovery into clinical trials,” said Jim Doroshow, MD, deputy director for Clinical and Translational Research at NCI.
Contributing authors on the NEJM publication are James E. Herndon II, Nike Beaubier, Dani P. Bolognesi, Allan H. Friedman, Henry S. Friedman, Frances McSherry, Andrea M. Muscat, Smita Nair, Katherine B. Peters, Dina Randazzo, John H. Sampson, Gordana Vlahovic, William T. Harrison and Roger E. McLendon.
The work received grant support from The Brain Tumor Research Charity, the Tisch family through the Jewish Communal Fund, Uncle Kory Foundation, Department of Defense (W81XWH-16-1-0354), and National Institutes of Health (R35CA197264, P01CA154291, P50CA190991, R01CA124756, R01NS099463). Support was also received through the Angels Among Us fundraising event and a gift from the Asness family. The NIH provided the foundational poliovirus to produce the therapy.
Top: Matthias Gromeier, MD, in his lab
Middle: Allan Friedman, MD, performing the catheter placement
Bottom: The poliovirus therapy will be placed directly into the tumor